����ҩʯ�Ƽ�����һ�����������������ɫ�����о��ɹ�����ʽ������������ѧ�ᣨACS����ɫ��ѧ��ɳ�����չר�������η����ijɹ�����Ϊ��˾���ɵ���ɫ�������켼��ƽ̨����ƽ̨����ں���ɫ��ѧ����ԭ������Ĺ�ҵ���������գ����¿�����Ӧ��������Һ����ĺϳɣ�CF-LPPS����������ϳɡ���ɫ�ܼ������ϵ������/Һ����ĺϳɹ����Ż���Ĥ��ѧ���ա��绯ѧ���յȹؼ�����������Դ�ͳ����������ܼ����ġ����ܺġ��������ɵ���ҵʹ��ʵ�ּ���ͻ�ƣ�����Ϊ��ҵ�������߹�ҵ�����������ԡ������Ѻ��ԵĹ�ģ������������·��������ʵ������ɫ��ѧ�����ڶ��Ĺ�ҵ�������е�������ҵ��ʵ����
������Ϊ����ҽҩ���²��ϵ�����ĺ��Ĺؼ�ԭ�ϣ��������칤�յ���ɫ����ɳ������ѳ�Ϊȫ������ҩ��ҵ�ĺ��ķ�չ���⣬����ͳ������DMF���ж��ܼ�������ԭ�Ӿ����Ե͡������ŷŸߵ����⣬ʼ����Լ����ҵ�ĵ�̼ת�ͣ�PTCoEij������Ŀͨ��LPPS��CF-LPPS������DMFʹ��������SPPS�IJ���ٷ�֮һ��ҩʯ�Ƽ�PTCoE�Ŷӳ��������һ���������켼���з����˴εǰ�ACS�ļ����ɹ��������ǹ�˾�ڷ���Ȼ�����ṩӦ�����ĺϳɡ���ɫ����һ�廯���ֵ���Ҫ���֣���ͨ��������ļ�����ЭͬӦ�ã���������˶��������Ĺ�������ǿ�ȣ�PMI����Ϊȫ�����������ҵ����ɫ�ɳ�����չ�ṩ�˿ɹ�ģ����ص�ʵ������������Ϊ����ԭ�ģ�����һͬ���������ڸ�����������̽����
����Peptides, as essential biomolecules linking amino acids and proteins, play a vital role across numerous disciplines, including life science research, drug discovery, biomaterials engineering, and diagnostic reagents development. Their value lies in their unique structural diversity, potent biological activity, and precisely controllable synthesis. At the heart of harnessing this molecular potential is peptide synthesis technology. Over the past century, this field has evolved from a complex laboratory process into a versatile and indispensable tool, blending scientific innovation with engineering precision. Today, peptide synthesis continues to drive advancements in biomedicine and materials science, unlocking new frontiers in both research and application.
����Green Chemistry and Sustainable Development for Peptides
����A common challenge confronting the peptide industry is that the transition toward green chemistry and sustainable development in peptide manufacturing constitutes a complex, multidisciplinary undertaking��a sophisticated systematic endeavor encompassing everything from efficient synthesis to purification processes. Traditional paradigms and methodologies for large-scale peptide production are proving increasingly insufficient to meet the stringent demands of green chemistry and sustainable development. Consequently, conducting sustained and in-depth research into peptide chemistry and exploring greener, more sustainable pathways for large-scale manufacturing represent an inevitable trajectory for the next generation of peptide production.
����PharmaBlock, a global Contract Research, Development, and Manufacturing Organization (CRDMO) company, and member of ACS GCI Pharmaceutical Roundtable, has established an integrated peptide synthesis technology platform that incorporates core methodologies such as, liquid-phase synthesis, enzymatic peptide synthesis, and continuous-flow peptide synthesis. These technologies can be flexibly combined based on the specific sequence and structural characteristics of the target peptide, thereby enabling a green and scalable supply of peptide-based therapeutics. PharmaBlock��s technologies were recognized two years in a row as winner of ACS GCIPR��s CMO Excellence in Green Chemistry Award in 2023 and 2024.

����Solid-Phase and Liquid-Phase Peptide Synthesis
����Solid-Phase Peptide Synthesis (SPPS), the predominant model, relies on large quantities of solvents and reagents. According to a recent study by the ACS GCI Pharmaceutical Roundtable, SPPS has a Process Mass Intensity (PMI) of about 13,000��significantly higher than over modalities. Compared to SPPS, Liquid-Phase Peptide Synthesis (LPPS) generally exhibits lower synthesis efficiency for long-chain peptides. However, as a homogeneous reaction system, LPPS offers irreplaceable advantages. LPPS operates in a homogeneous solution and eliminates costly solid supports, resulting in substantial cost savings during large-scale production. Unlike resin-based SPPS, LPPS is not limited by resin loading capacity or swelling constraints, allowing synthesis to be scaled up readily to kilogram quantities. Additionally, LPPS avoids the repetitive washing steps associated with solid supports, significantly reducing solvent consumption��an important environmental and economic benefit at industrial scales. In LPPS, each synthesis step occurs in solution, allowing intermediates to be isolated, purified and characterized using conventional separation techniques. This enables effective removal of unreacted starting materials, by-products, excess reagents. Purifying intermediates after each step yields final crude products with higher purity than that obtained via SPPS, reducing final purification costs and burden. Moreover, the ability to characterize intermediates at every stage ensures structural accuracy and synthesis fidelity. This control is critical in the production of high-value or structurally complex peptides, such as active pharmaceutical ingredients (APIs), where precision, reproducibility, and regulatory compliance are paramount.
����For short peptides (typically 2�C10 amino acids), LPPS provides a relatively straightforward workflow, easier purification, and lower costs compared to SPPS. The homogeneous solution environment in LPPS is particularly advantageous for intramolecular cyclization reactions, enabling better reaction monitoring and control over cyclization conditions. This makes LPPS the preferred method for synthesizing head-to-tail or side-chain cyclic peptides. Additionally, purifying linear peptide precursors before cyclization enhances both cyclization efficiency and final product purity.
����LPPS serves as the primary platform for fragment condensation, overcoming the challenges of low efficiency and impurity accumulation in SPPS for long peptide chains. In LPPS, unnatural amino acids and complex modifications can be introduced into fragments with high efficiency and purity. Following purification and structural confirmation, these well-characterized segments are condensed to form the full-length peptide sequence, significantly enhancing overall yield and final product purity. Moreover, multiple fragments can be synthesized in parallel and subsequently assembled, dramatically reducing the synthetic time for long peptides compared to conventional linear SPPS. A hybrid approach��using SPPS for fragments production and LPPS for fragment condensation��further enhances synthetic efficiency, combining the strengths of both methods.
����Innovation in LPPS: Continuous Flow Liquid-Phase Peptide Synthesis (CFLPPS)
����CFLPPS technology combines the scalability and flexibility of LPPS with the precision and enhanced mass-transfer capabilities of microreactors. This integration improves reaction efficiency, safety, and process control while effectively addressing scalability, and environmental challenges associated with conventional batch methods. As a result, CFLPPS offers a promising avenue for sustainable, high-throughput production of peptides with improved reproducibility and reduced waste.
����CFLPPS facilitates high-throughput screening with minimal material consumption, allowing rapid optimization of critical parameters such as temperature, concentration, and flow rate. This drastically shortens route development timelines compared to conventional LPPS. Importantly, once optimized, scaling up production can be achieved seamlessly by increasing the number of parallel microreactors, enabling smooth transition from milligram scale laboratory synthesis to kilogram or even ton-scale manufacturing. This approach eliminates traditional scale-up challenges, ensuring consistent product quality and process reliability, which are crucial factors for active pharmaceutical ingredient (API) production.
����CFLPPS is amenable to real-time monitoring and full automation. The precise fluid control by high-precision injection pumps ensures that all reactants are delivered to the microreactor at precisely defined stoichiometric ratios and constant flow rates, eliminating human error and concentration gradients often observed in batch processes. Besides, software-driven operation removes manual intervention variability, ensuring controlled and reproducible synthesis conditions. Furthermore, CFLPPS can be fully automated, with all stages��including reaction, quenching, extraction, and separation��integrated into a modular, continuous production workflow. This automation lays the foundation for intelligent peptide manufacturing and ensures robust inter-batch consistency, scalability, and regulatory compliance in pharmaceutical production.
����The automation of CFLPPS generates rich real-time data streams that can be effectively harnessed by artificial intelligence (AI) and machine learning (ML) models. These algorithms can automatically analyze process data, dynamically adjust reaction parameters, and autonomously identify optimal operating conditions in real time. By aggregating and interpreting large datasets, digital twins of the process can be developed for simulation, optimization, and operator training��reducing both labor and resource requirements. Coupling AI systems with online monitoring enable continuous status updates and feedback control, facilitating autonomous decision-making and real-time self-optimization. This integrated approach delivers unprecedented levels of process control and production efficiency, establishing a transformative paradigm for the future of peptide manufacturing.
����Hybrid Processes and Enzyme-Assisted Peptide Synthesis
����Compared to fully SPPS, conventional hybrid processes can substantially reduce the PMI in the production of long-chain peptides such as GLP-1 analogs, while also facilitating more effective quality control. Nevertheless, current mainstream fragment ligation strategies remain predominantly reliant on chemical methods. This approach necessitates a secondary cleavage step following ligation, as the initial cleavage occurs during the solid-phase synthesis stage. The subsequent purification requirements introduce considerable challenges and leave substantial room for improvement from a green chemistry perspective. Pushing process boundaries beyond fully optimized parameters is essential in the pursuit of green chemistry and sustainable development for peptide manufacturing. Enzyme-Assisted Peptide Synthesis (EAPS) may serve as a valuable complement to hybrid strategies in this regard.
����PharmaBlock has constructed an enzyme library comprising multiple species of peptide ligases. Experimental results demonstrate that employing peptide ligases in the synthesis of long peptide fragments enables precise assembly of unprotected peptides. The reaction proceeds in aqueous medium, and the final product can be isolated through centrifugation and membrane filtration without requiring secondary cleavage, thereby significantly reducing PMI. Looking ahead, the application of peptide ligases could evolve toward immobilized enzyme systems, further advancing EAPS toward a continuous production mode. With respect to both quality control and downstream purification requirements, this production paradigm represents a qualitative leap, enabling substantial PMI reduction in large-scale peptide manufacturing.
����Cyclic peptides are progressively emerging as a key direction for future peptide research and development. Achieving green chemistry and sustainable development in the cyclic peptide arena is therefore a critical consideration for large-scale peptide manufacturing. In this context, LPPS and CFLPPS represent important technological avenues for addressing the challenges associated with cyclic peptides. A cyclic peptide project from PharmaBlock��s broad project portfolio provides a valuable reference. Initially, an SPPS route was adopted for the first synthesis of this cyclic peptide. This approach involved extensive use of DMF as a solvent during production, and the final product was obtained through reversed-phase chromatography purification, with a maximum batch size of approximately 500 g. However, as the required supply scale increased and cost pressures mounted, the SPPS route became economically and environmentally unsustainable. Consequently, an LPPS process was developed to enable large-scale production. Implementing the LPPS process reduced DMF usage to less than 1/20 of that required for SPPS. Furthermore, purification via reversed-phase chromatography was effectively circumvented through crystallization of key intermediates. Nevertheless, there remained significant room for optimizing the PMI of the post-treatment steps in certain segments of the LPPS process, making the pursuit of greener methodologies a key objective.
����Building upon the LPPS process for this cyclic peptide, PharmaBlock developed a CF-LPPS process. By integrating continuous reaction, continuous work-up, membrane-based separation, and process analytical technology (PAT), fully continuous synthesis of the linear peptide was achieved. Following crystallization of the linear peptide, the cyclization step was performed using a microchannel reactor and a continuous stirred-tank reactor (CSTR), effectively enabling the CF-LPPS synthesis of the cyclic peptide. Through this process evolution��from SPPS to LPPS and finally to CF-LPPS for the cyclic peptide��DMF consumption was reduced to less than 1/100 of that in the original process. Moreover, the need for reversed-phase chromatography preparation was eliminated, avoiding the use of large quantities of acetonitrile and the lyophilization step, thereby rendering the cyclic peptide synthesis substantially greener and more sustainable.
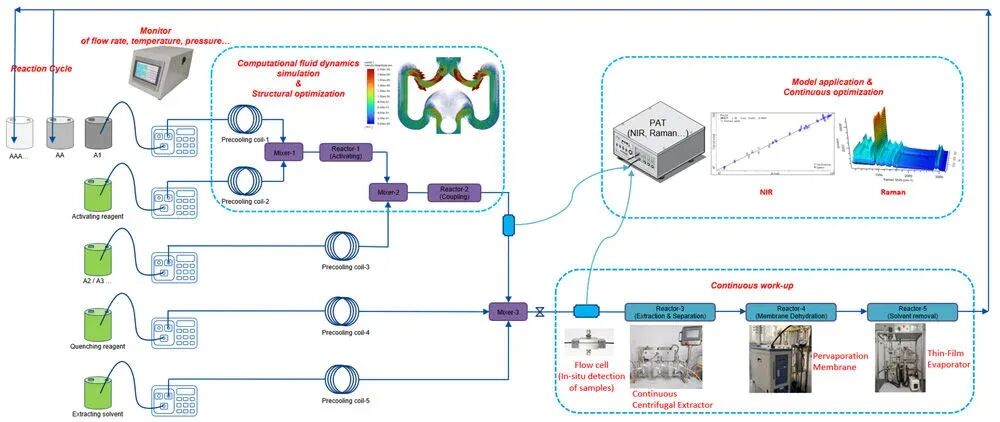
����Picture 1. Set up of CF-LPPS for a cyclic peptide
����Green Chemistry and Sustainable Development in the peptide field necessitate not only continuous exploration of synthetic processes but also represent a future research direction focused on achieving further advancements in peptide purification and post-treatment processes. Traditional peptide purification primarily relies on reversed-phase chromatography, which involves substantial acetonitrile usage and generates astronomically large volumes of waste relative to the product yield. Apart from certain peptides amenable to crystallization purification, enhancing the efficiency of reversed-phase chromatography purification and controlling acetonitrile consumption constitute an essential pathway for green peptide production. Multi-column counter-current solvent gradient purification (MCSGP) integrates the counter-current operational principle of simulated moving bed technology with solvent gradient elution techniques, achieving continuous purification through the coordinated operation of multiple chromatographic columns. Reports indicate that this approach has successfully completed PPQ validation for commercial production. Compared to conventional batch chromatography, MCSGP increases purification yield by 20% to 30%, enhances production efficiency by 20 to 25-fold, and reduces PMI by 70%.
����Beyond advancements in purification technology, effective treatment of purified solutions represents a major area of development and concern. Column chromatography for concentration and salt exchange constitutes the most common post-treatment approach for purified solutions; however, this method still requires substantial quantities of organic solvents such as acetonitrile. The application of membrane chemistry enables direct concentration of prepared solutions without chromatographic columns, eliminating additional solvent requirements during the concentration step. For salt exchange, electrochemical approaches offer viable solutions. In actual production cases at PharmaBlock, certain GMP manufacturing projects have implemented integrated post-treatment solutions combining membrane chemistry with electrochemical approaches to replace traditional column chromatography concentration and salt exchange methods. This integration has increased single-step yield from 60% to 90%, reduced single-step costs to merely one-tenth of the original process, and compressed production timelines to one-fifth of the original duration.
����PharmaBlock��s Commitments to Green, Continuous Peptide Manufacturing
����PharmaBlock is dedicated to establishing a green, low-carbon, and continuous peptide manufacturing platform. The company provides integrated services spanning from small-scale development to GMP-compliant production. By enhancing its liquid-phase peptide synthesis (LPPS) capabilities through advanced continuous flow and biocatalysis platforms, PharmaBlock is actively building robust infrastructure to meet large-scale peptide delivery needs. In the long term, the company is focused on developing CRDMO platform for peptides based on CFLPPS and biocatalysis��merging innovation, sustainability, and scalability to redefine the future of peptide therapeutics.
�����Ͼ�ҩʯ�Ƽ��ɷ�����˾����Ʊ���룺300725����˾��ƣ�ҩʯ�Ƽ�����ȫ��ҽҩ�з������������»�ѧ��Ʒ�ͷ���Ӧ�̡���˾ʼ��������ͨ���з������������еĻ�ѧ�͵�̼�����Ĵ��£�����������������ҩ���ּ�����Ч�ʣ�ȷ����Ʒ�������ȶ������������з��������ɱ����������ƶ���ҵ����ɫ���ɳ�����չ��
������2008��Ͷ����Ӫ������ҩʯ�Ƽ��ѳɹ���ȫ����������ǰ��ʮ����ҩ��˾����ǧ����С�����\����˾��ɺ�����ҩʯ�Ƽ���ҵ��Դ������ӱ�������Ҿ���ǰհ�Եķ������飬��Щ���������ҩ��ֵ��ƽ��ؼ����ã�����������Щ��������ĺ�ѡ����������ٴ���������ҵ���Σ�ҩʯ�Ƽ��ɹ���������ǧ�ַ��������Ʒ�ķŴ��գ���ʵ�ֹ�ģ���������Ӷ������ͻ�Ѹ���ƽ�ҩ�↑����Ŀ��ƾ���ڷ�����������������ۡ��ͻ��ĸ߶��������Լ�����ԭ�Ϲ�Ӧ�ͻ�ѧ����ȶ������ƣ�ҩʯ�Ƽ�������չҵ������Ϊҩ����ṩ��ѧ�з�����Ϊ�ٴ�ǰ���ٴ���������ҵ����Ŀ�ṩ��Ч����Ʒ�ʵ��м��塢ԭ��ҩ��ҩ���Ƽ��Ĺ��տ�������������ͬʱ����˾���϶���������������ѧ����䴲�����������������������ǰ�ؼ����ϵ��������ۣ�����̽������ҽҩ������ɫ����ȫ�����ܻ����Ƚ����켰����ģʽ���ٽ���ҵ���·�չ��